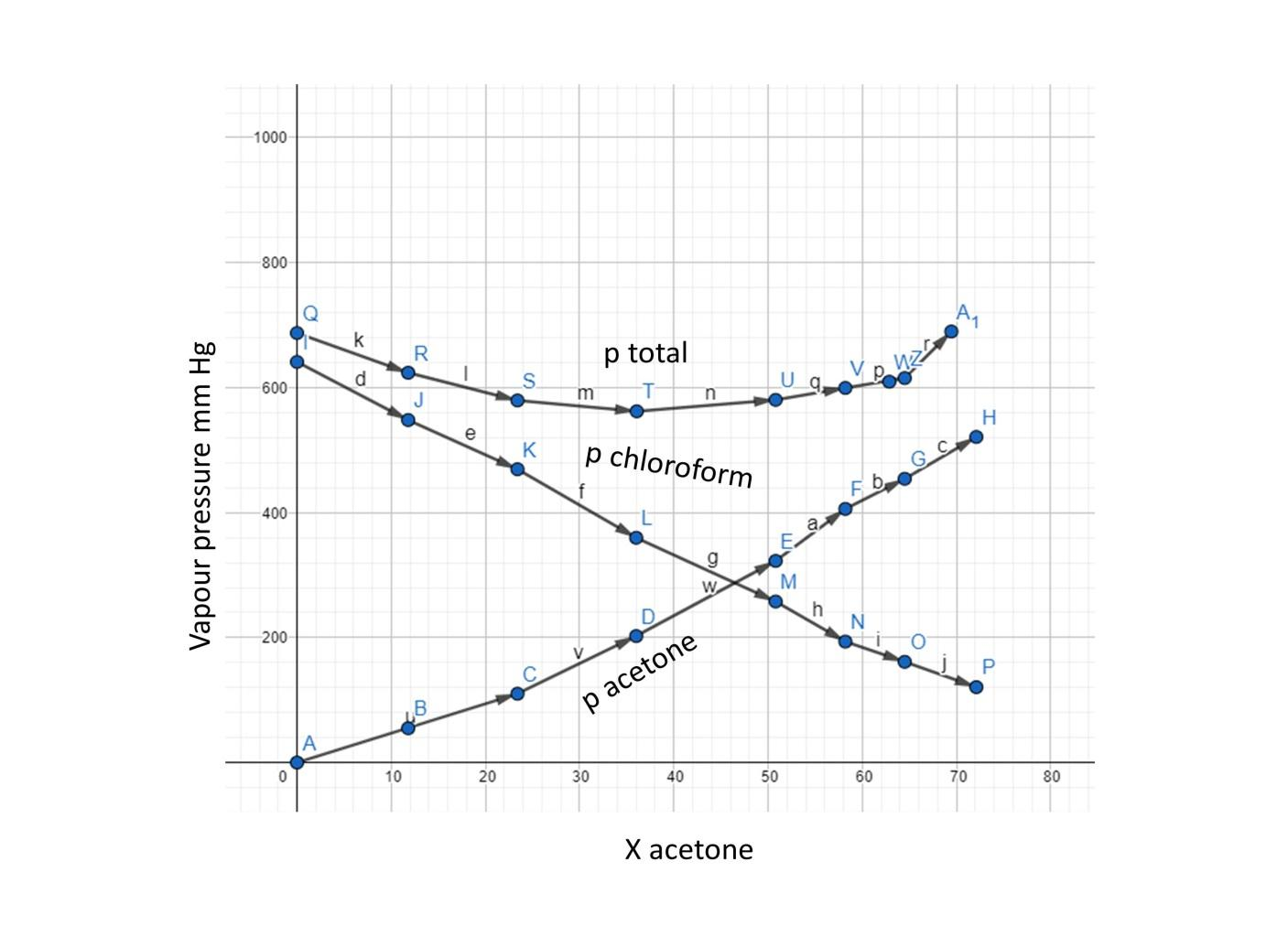
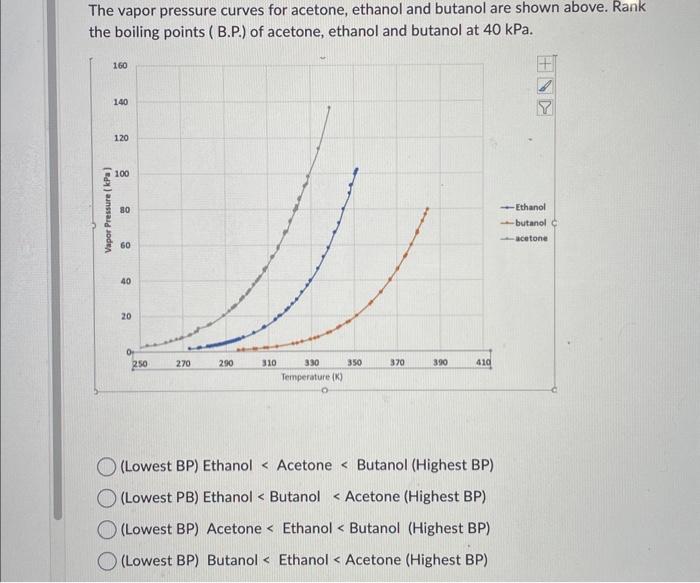
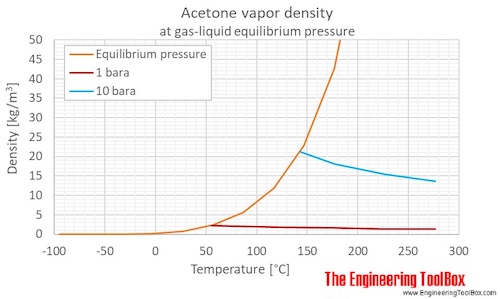
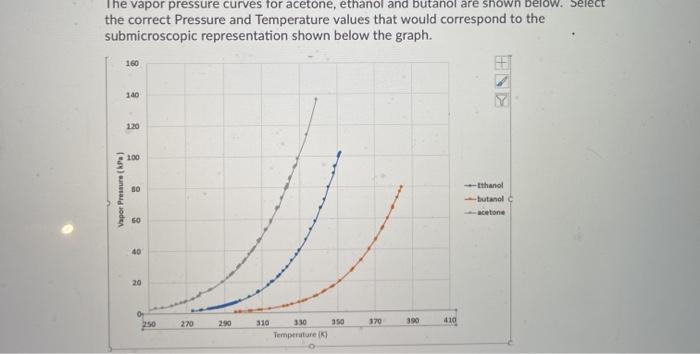
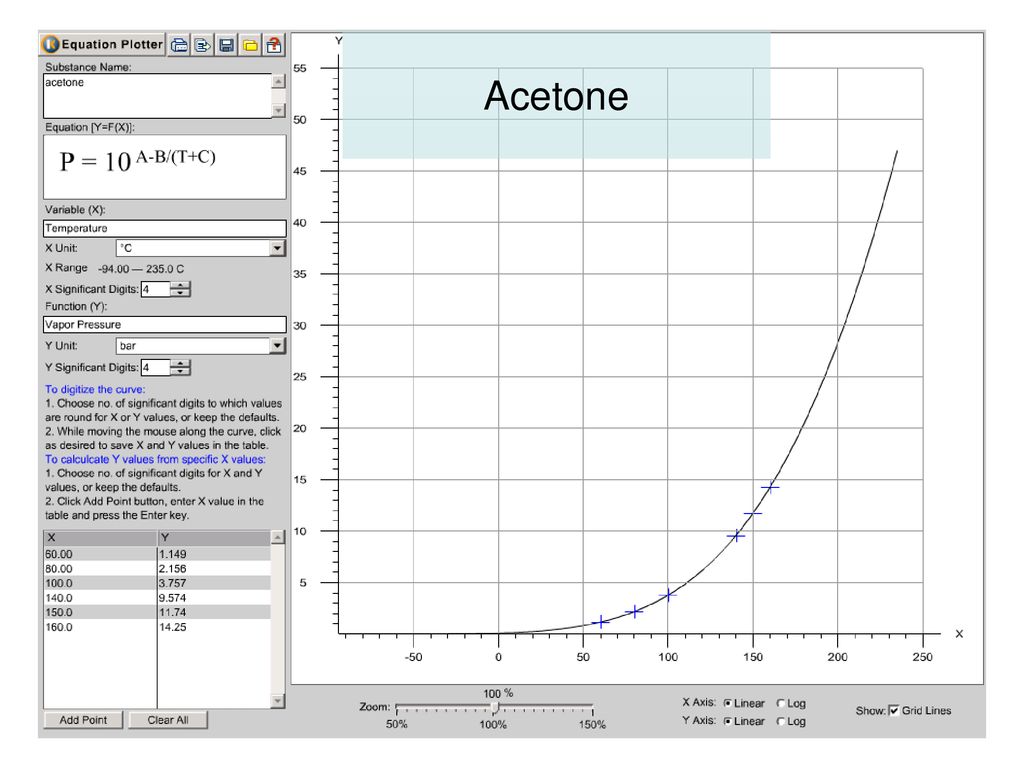
QUESTION 2.37 Vapour pressures of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of

Use the following vapor pressure diagram to estimate the partial pressure of acetone. | Homework.Study.com

The saturation vapor pressure of the benzene/acetone mixture (C 6 H 6 +... | Download Scientific Diagram
Isobaric vapor-liquid equilibrium for acetone + methanol system containing different ionic liquids at 101.3 kPa
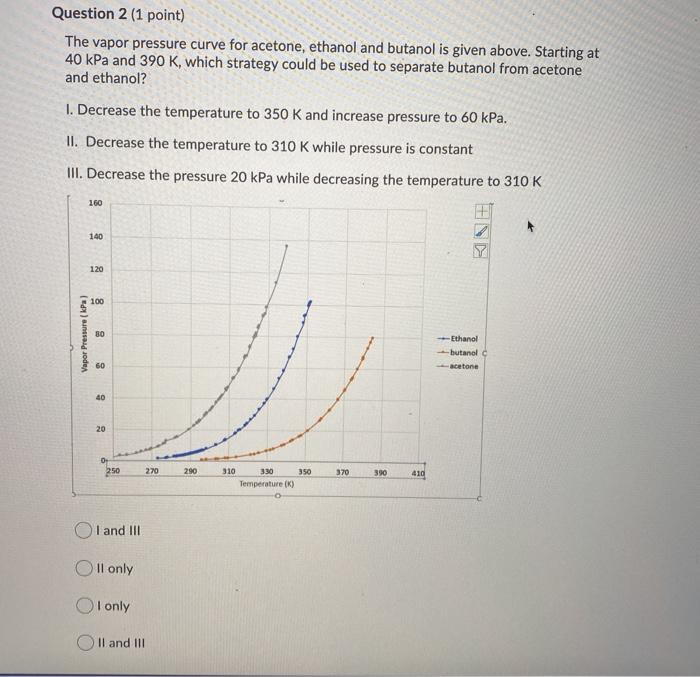
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

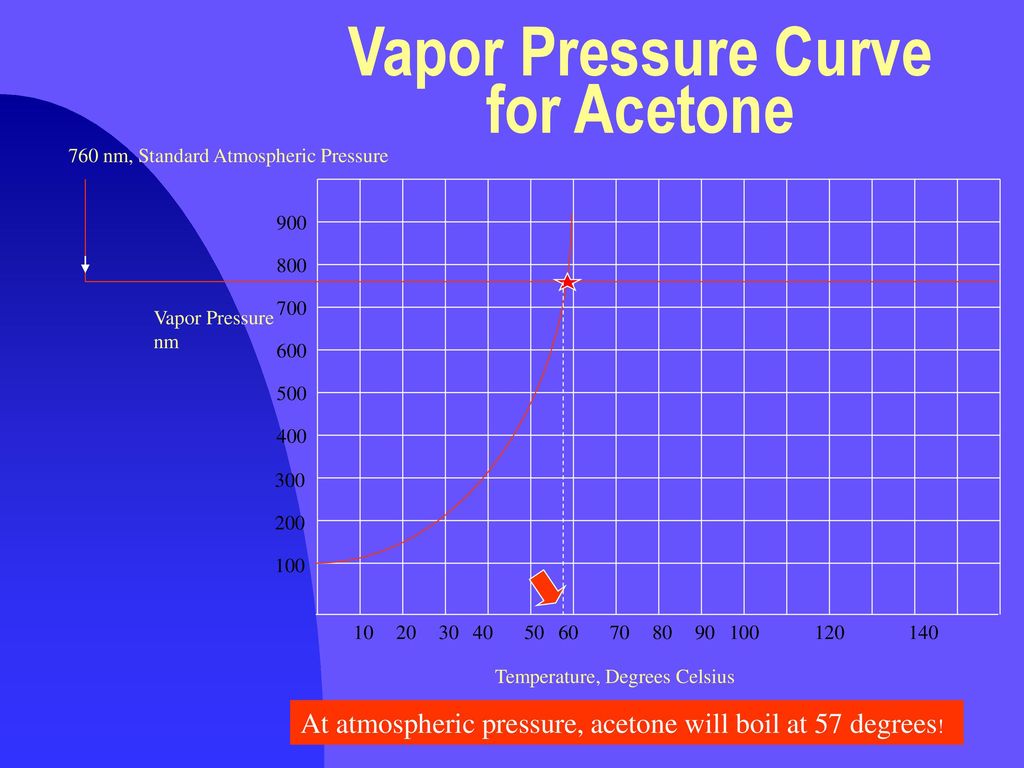
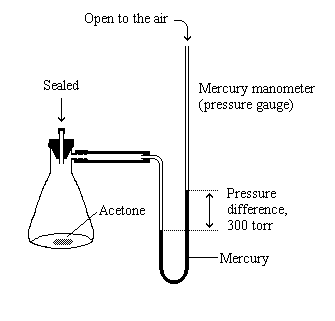
The vapor pressure of acetone at `20^(@)C` is 185 torr. When `1.2 g` of a non-volatile solute - YouTube

Vapour pressure of pure acetone and chloroform at 328 K are 741.8 mm Hg and 632.8 mm Hg respectively. Assuming that they form ideal solution over the entire range of composition, plot